 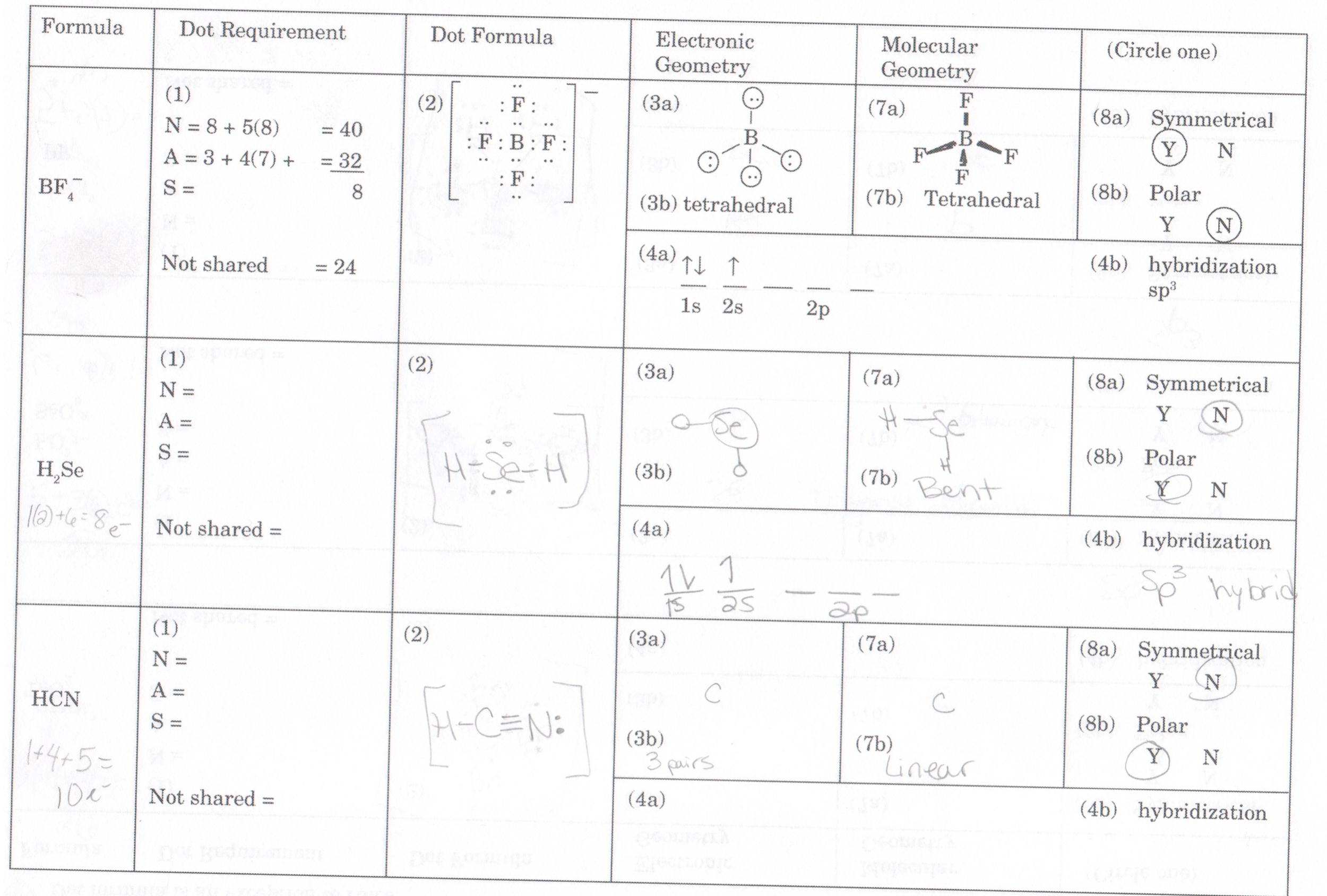
Next, a skeletal structure is formed by determining the central atom(s). Phosphorus Pentachloride possesses forty valence electrons, as calculated in the previous section. 
Therefore, the total number of valence electrons made available by Phosphorus Pentachloride (PCl 5) is given by:ĥ + 35 = 40 valence electrons Lewis Structure Assembly Therefore, the five Chlorine atoms present contribute 7 x 5 = 35 valence electrons. The electronic configuration of Chlorine is given by 3s 23p 5. Therefore, the single Phosphorus atom in Phosphorus Pentachloride contributes 5 x 1 = 5 valence electrons.īeing in group 7 of the periodic table, Chlorine has seven valence electrons. Phosphorus is present in group 5 of the periodic table of elements with the electronic configuration: 3s☣p³. Let us now determine the number of valence electrons present in this molecule. PCl 5 comprises a single Phosphorus atom and a set of five Chlorine atoms. The available valence electrons are used to build and define the Lewis structure. This position enables valence electrons to break free to participate in bond formation.ĭepending upon the atomic number and position of the element on the periodic table, a set number of valence electrons are made available. They lie in the atom’s outermost shell, where the force of attraction tethering them to the nucleus is at its weakest. Valence electrons participate in covalent bonding and electron exchange by virtue of their position with respect to the atomic nucleus. Valence ElectronsĮach electron in an atom is accounted for and can be found in particular orbitals and subshells depending on their quantum properties. The first step in obtaining the Lewis structure for a particular molecule is to calculate the number of valence electrons. Lewis structures also give insight into the chemical polarity, molecular geometry & bond angles of a molecule. The structure uses elementary dots and lines to describe bonds between atoms and valence electrons present. Concluding Remarks PCl 5 Lewis StructureĬhemical bonds and electrons present in a molecule can be accurately represented using schematic diagrams called Lewis structures.of valence electrons (5 x 1) + (7 x 5) = 40 valence electrons Hybridization of the central atom sp 3d Bond Angles 90° & 120° Molecular Geometry of PCl 5 Trigonal Bipyramidal The physical properties of Phosphorus Pentachloride (PCl 5) are given below: Name of the molecule Phosphorus Pentachloride (PCl5) No. PCl 5 is a precursor for components used to manufacture Lithium-ion batteries. PCl 5 is electrophilic and is also a Lewis acid. Carboxylic acids can be converted to acyl chloride, whereas alcohols can be converted to alkyl chlorides. PCl 5 is used in both oxidative and substitutive chlorination reactions. PCl 5 undergoes hydrolysis to release hydrogen chloride and oxides of phosphorus. Phosphorus Pentachloride is prepared by the dry action of Chlorine on PCl 3 in a chlorination reaction. It partially dissociates after self-ionization in polar solvents such as Nitrobenzene. In a solid state at room temperature, PCl 5 exists as an ionic compound with the formula PCl + 4PCl – 6. 
It is mainly used as a chlorinating reagent. It is a greenish-yellow crystalline solid with an irritating odor. The chemical formula PCl 5 represents the chemical compound Phosphorus Pentachloride. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
